
It is higher in eukaryotes than prokaryotes and in multicellular than unicellular organisms. 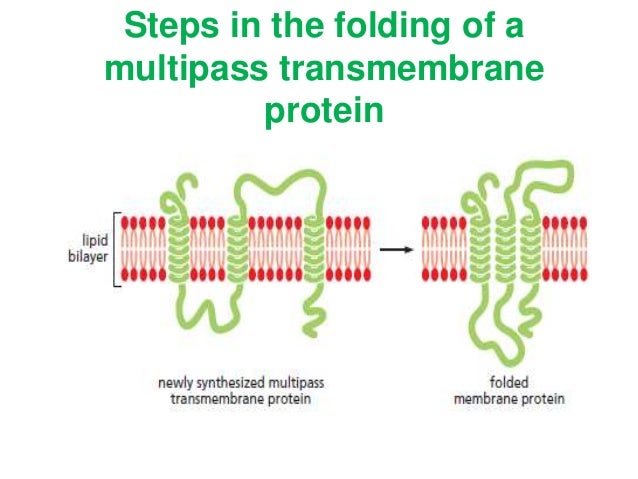
The number of single-pass transmembrane proteins in an organism's genome varies significantly. Some proteins in this class function as monomers, but dimerization or higher-order oligomerization is common. The ECD is typically much larger than the ICD and is often globular, whereas many ICDs have relatively high disorder. The transmembrane domain is the smallest at around 25 amino acid residues and forms an alpha helix inserted into the membrane bilayer. Type II and III are anchored with a signal-anchor sequence, with type II being targeted to the ER lumen with its C-terminal domain, while type III have their N-terminal domains targeted to the ER lumen.Ī single-pass transmembrane protein typically consists of three domains, the extracellular domain, the transmembrane domain, and the intracellular domain. Hence type I proteins are anchored to the lipid membrane with a stop-transfer anchor sequence and have their N-terminal domains targeted to the ER lumen during synthesis. Type IV: N-terminus on the cytoplasmic side of the membrane transmembrane helix located close to the C-terminus, where it works as an anchor.Type III: N-terminus on the extracellular side of the membrane no signal peptide.Type II: N-terminus on the cytoplasmic side of the membrane transmembrane helix located close to the N-terminus, where it works as an anchor.Type I: N-terminus on the extracellular side of the membrane removed signal peptide.Topology-based classification īitopic proteins are classified into 4 types, depending on their transmembrane topology and location of the transmembrane helix in the amino acid sequence of the protein. A 2013 estimate identified about 1300 single-pass membrane proteins in the human genome. 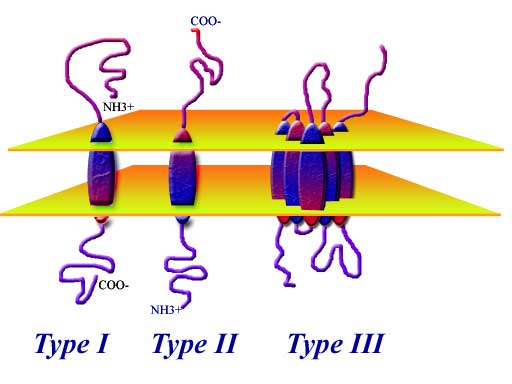
Some of them are small and serve as regulatory or structure-stabilizing subunits in large multi-protein transmembrane complexes, such as photosystems or the respiratory chain. They usually include one or several water-soluble domains situated at the different sides of biological membranes, for example in single-pass transmembrane receptors. These proteins may constitute up to 50% of all transmembrane proteins, depending on the organism, and contribute significantly to the network of interactions between different proteins in cells, including interactions via transmembrane alpha helices. a polytopic transmembrane β-sheet proteinĪ single-pass membrane protein also known as single-spanning protein or bitopic protein is a transmembrane protein that spans the lipid bilayer only once. a polytopic transmembrane α-helical protein 3. a protein with single transmembrane α-helix (a single-pass membrane protein) (bitopic) 2. Schematic representation of transmembrane proteins: 1.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
